Description
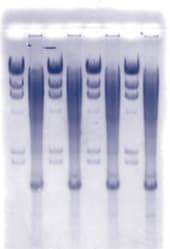
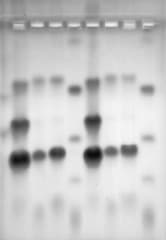
Purified proteins are often needed in the basic research laboratory and for diagnostic and therapeutic procedures. An effective technique for protein purification is affinity chromatography, which exploits a specific interaction between a protein and a complementary binding molecule. In this exercise, students isolate albumin from horse serum by affinity chromatography using a column matrix containing a reactive blue dye which binds specifically to the albumin molecule. They then use electrophoresis to analyze the isolated protein in order to verify the effectiveness of the procedure. This exercise requires approximately two 2-3 hour laboratory periods and the materials provided to perform the experiment are shown.
Each of the individual experiments are supplied with the chemicals and laboratory guides needed for 16 students working in pairs. If you chose one or more of the experiments from this series – you should also order Electrophoresis Package 2M or Electrophoresis Package 2. Electrophoresis Package 2M provides sufficient agarose, gel stain, and electrophoresis buffers for 1 of the individual experiments in this series (four gels with 15ml of agarose per gel). Electrophoresis Package 2 provides sufficient agarose, gel stain, and electrophoresis buffers for up to 6 of the individual experiments in this series.
If ordering the EXP-206P variant, precast polyacrylamide gels are not included. Precast polyacrylamide gels can be ordered from Bio Rad, (800-424-6723) Their catalog number is 4561093EDU.
 Due to Customs restrictions, we only accept orders from educational institutions within the Continental United States, Alaska or Hawaii.
Due to Customs restrictions, we only accept orders from educational institutions within the Continental United States, Alaska or Hawaii.